CAMPBELL, Calif., Sept. 21, 2022 /PRNewswire/ -- Vivalink, a leading provider of digital healthcare solutions, has released an updated version of its Biometrics Data Platform with new features aimed at increasing patient adherence in Remote Patient Monitoring (RPM).
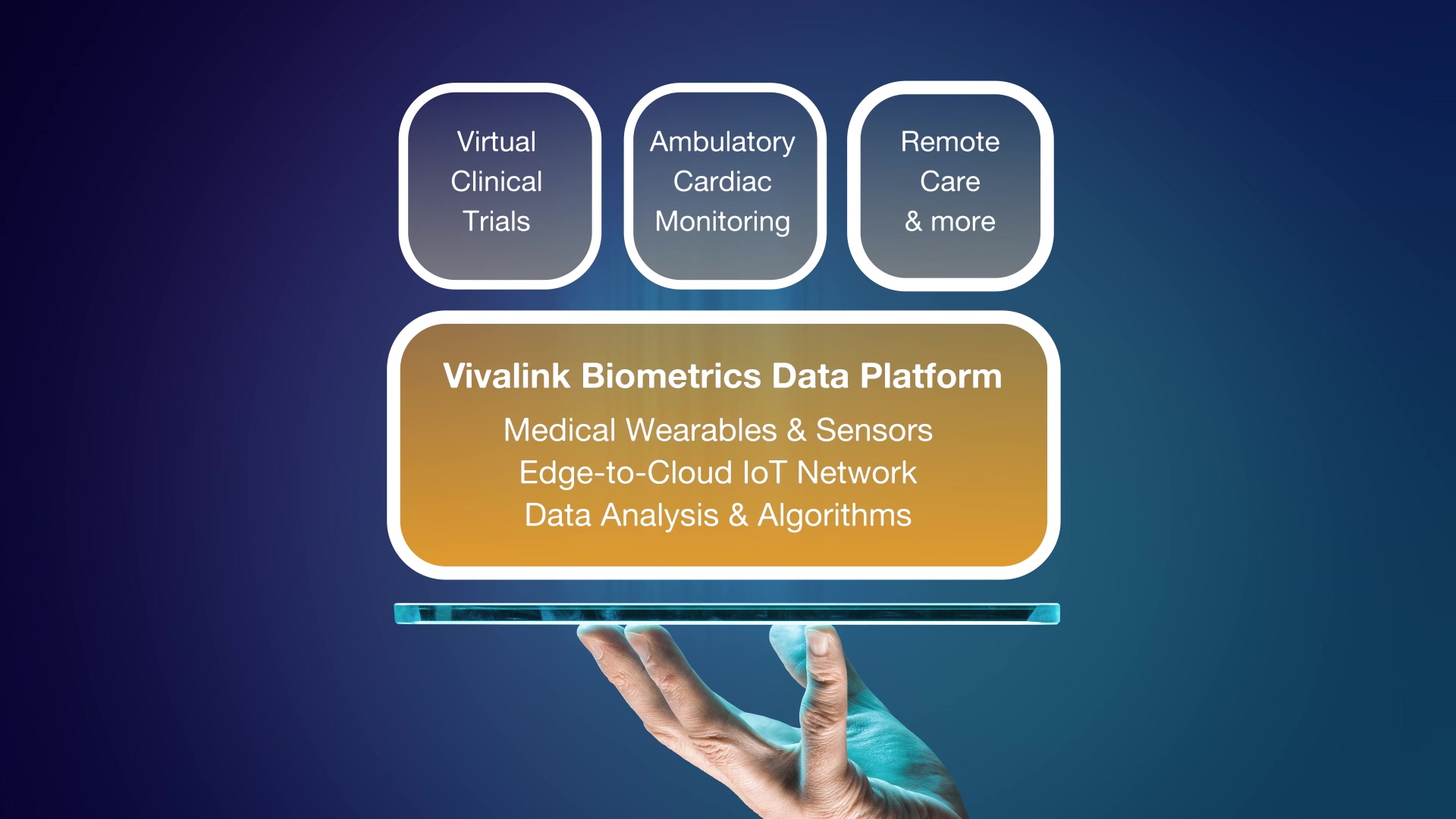
Vivalink's Biometrics Data Platform accelerates the implementation and time-to-market of remote patient monitoring for healthcare and clinical trials by combining a turn-key end-to-end solution consisting of a suite of wearable sensors, edge-to-cloud integration, and advanced data management and analytics both in real-time and retrospectively. The software platform is designed for flexible interoperability with mobile and web enabled clinical applications, electronic health record (EHR) systems and clinical trial management systems (CTMS).
While the adoption of RPM has accelerated with the pandemic and is expected to grow exponentially, patient adherence remains a top challenge. In a Decentralized Clinical Trial (DCT) survey by Vivalink, 69 percent of respondents indicated 'patient adherence' as the top concern.
To address this concern, the new Biometrics Data Platform updates include:
Vivalink takes a "patient centric" approach to its RPM solution, which includes making its devices as easy to use and comfortable as possible for the patient. For example, its medical grade multi-vital cardiac patch is among the world's smallest and lightest, weighing just 7.5 grams, increasing comfort while reducing disruption to daily routines
"As a technology solution provider, we strive for the most advanced and effective solutions for remote patient monitoring," said Jiang Li, CEO of Vivalink. "Patient adherence is a major bottleneck for the wider adoption of RPM. Today's release of these features focusing on improving patient adherence shows our firm commitment to the patient-centric approach of our solution."
More than 150 digital healthcare solution partners and commercial customers in 41 countries are delivering a wide range of medical applications using Vivalink's Biometrics Data Platform. Vivalink's solution is the basis of clinical research studies at leading institutions, including a 3,000 patient atrial fibrillation study at UCSF and a study of the effects of stress and teenage depression at Stanford University. Most recently, the platform has been adopted by health technology companies developing AI solutions, algorithm development and validation. The platform meets high levels of global compliance, including HIPAA, GDPR, FDA 21 CFR Part 11, ISO 13485, IEC 62304, ISO 14971, ISO 10993, and ISO 27001
Media Contact:
vivalink@ampublicrelations.com
VivaLNK, Inc.
51 E. Campbell Avenue, Suite 160
Campbell, California 95008
Sales: info@vivalink.com
Support: support@vivalink.com
No Comments Yet
Let us know what you think